Mastery – Molar Mass from Boiling Point or Freezing Point
OWLv2 for General Chemistry
Cengage
01st Edition
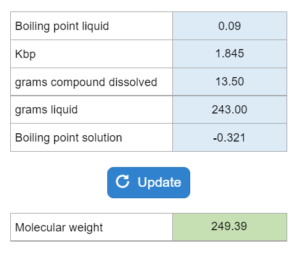
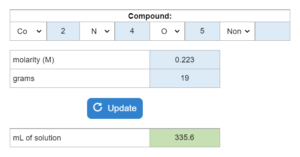
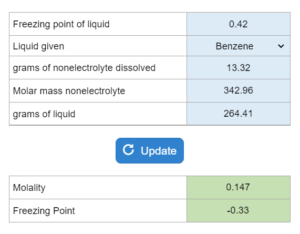
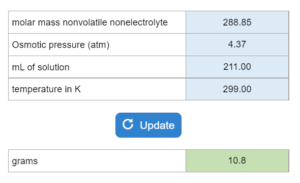
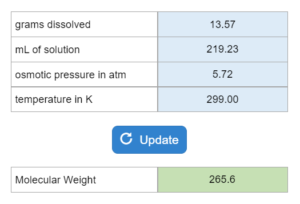
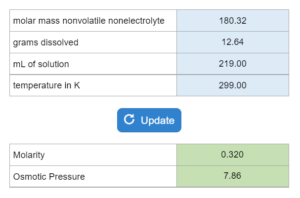
The boiling point or freezing point of a liquid is given as well as the Kb. X grams of an unknown compound are dissolved in x grams of liquid creating a new boiling or freezing point. Calculate molecular weight for the unknown compound. NOTE: this solver works whether you are given boiling points OR freezing points.