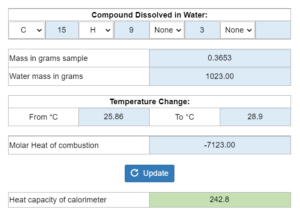
Mastery – Bomb Calorimetry: Calculate Heat Capacity of Calorimeter
A student burns x grams of a sample in a bomb calorimeter with x grams of water causing a temperature change. Molar heat of combustion and a balanced reaction are given. What is the heat capacity of the calorimeter?
Experts Have Solved This Problem
Please login or register to access this content.