Tutor – Predicting Evaporation or Condensation Using Vapor Pressure
OWLv2 for General Chemistry
Cengage
01st Edition
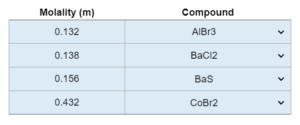
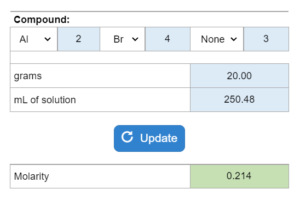
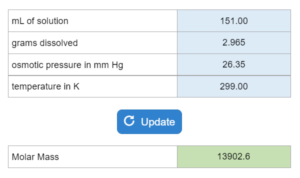
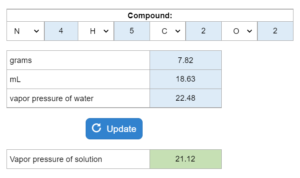
Choose all that apply. A liquid’s vapor pressure (in mmHg)at a temperature (K) is given. The compound is placed in a container (mL) and put at a certain pressure (mmHg). If the container is reduced to a new mL, what happens?